|
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The C2H4 bond angle will be about 120 degrees since it has a trigonal planar molecular geometry. The Carbon on the right will also have a trigonal planar molecular shape. Ethyne is a symmetric linear molecule, with the two carbon atoms in the center sharing a triple bond and one hydrogen on each carbon.Īs a result they will be pushed down giving the C2H4 molecule a trigonal planar molecular geometry or shape with respect to the Carbon on the left. Calculate the total number electrons around the central atom.Įthyne, which has the formula C2H2, is a nonpolar molecule.Determine the number of electrons donated by other atoms.Determine the number of valence electrons in the central atom.Predict the central atom of the molecule. Acetylene forms an explosive gas in the presence of air. The common name for this molecule is acetylene. 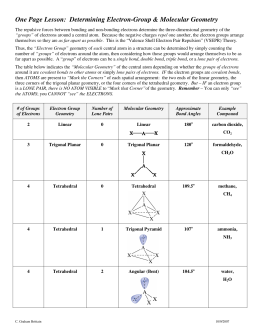
What is the electronic geometry of C2H2?Ĭ2H2 has a straight-line molecular geometry consisting of a hydrogen atom bonded to a carbon atom, which is triple-bonded to a second carbon atom bonded to a second hydrogen atom. Remember that, there are total of five electron pairs to mark on atoms. Mark lone pairs on carbon and hydrogen atoms After deciding the center atom and basic sketch of C2H2 molecule, we can start to mark lone pairs on atoms. For, ethene molecule, Total number of pairs of electrons are 6. Total electron pairs are determined by dividing the number total valence electrons by two. Looking at the positions of other atomic nuclei around the central determine the molecular geometry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
